News

The Kentucky Research Data Center (KRDC) is now open on UK’s campus. The facility offers researchers with approved projects the opportunity to access restricted data from the Census Bureau, the Bureau of Labor Statistics, and the Centers for Disease Control and Prevention. The Center is located in Suite 125A of the Gatton College of Business and Economics. Because the KRDC is a high security federal statistical research data center, those interested in conducting work at the facility must undergo a project review and obtain security clearance to access the Center.


Several new features will be added to xTRACT in a software release scheduled for Thursday, August 24, 2017. xTRACT is the Extramural Trainee Reporting and Career Tracking system and is accessed via eRA Commons. It allows applicants, grantees and assistants to create research training tables for progress reports and institutional training grant applications.

The Graduate School Tuition Fee Schedule was updated recently. The FY 2017/2018 acutal universal tuition rate for a full-time GA/RA is $12,246 ($6,123 per semester); and the actual health insurance premium is $2,166 ($823 – fall semester, and $1,343 – spring semester).
For more information, click here.

Consistent with the University of Kentucky’s (UK) mission, vision, and values, the Office of the Vice President for Research (VPR) provides programs and services to facilitate research, as well as support UK faculty, staff and students in research development and submission.

The UK Proposal Development Office (PDO) is a pre-award resource within the suite of services provided by the Office of the Vice President for Research to support strategic research development across campus through targeted resource and service delivery. PDO provides an array of services to faculty, staff, and students to assist them in securing extramural funding for scholarly activities. Primary research development services include:
coordination of complex multi-disciplinary projects and proposals;

[from the NIH website]
Message to NIH grant applicants/awardees, contractors, researchers and research administrators:
If you are conducting NIH-funded research that involves human subjects, or are considering applying to NIH for support of such research, we want to call your attention to important changes that may affect how you:

On August 2, 2017, NIH released an updated policy eliminating appendix material related to clinical trials for applications submitted to the NIH, AHRQ or NIOSH for due dates on or after January 25, 2018. The new policy specifies that all information for the peer review process must be contained within the application itself, in the designated section.
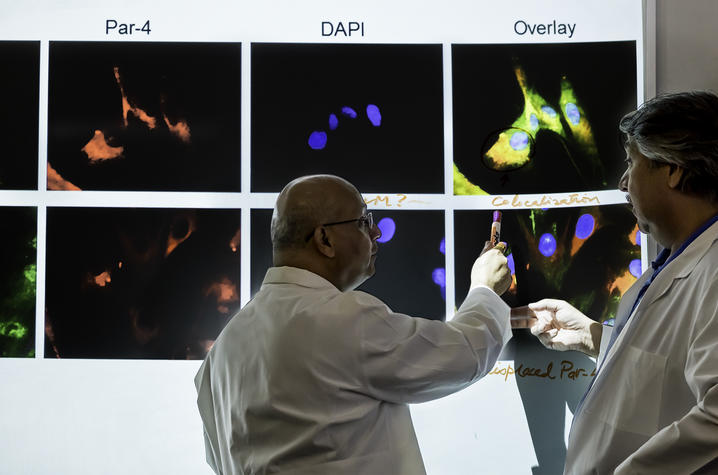
A new study by University of Kentucky Markey Cancer Center researchers shows that when therapy-sensitive cancer cells die, they release a "killer peptide" that can eliminate therapy-resistant cells.
Tumor relapse is a common problem following cancer treatment, because primary tumor cells often contain therapy-resistance cancer cells that continue to proliferate after the therapy-sensitive cells have been eliminated.

NIH recently updated the RPPR FAQs page, specifically related to Final RPPRs. Please review the FAQs here.

[From the NIH Website]
Additional Guidance on ‘Full-Time Training’ for Ruth L. Kirschstein National Research Service Awards
Notice Number: NOT-OD-17-095
Key Dates
Release Date: July 24, 2017
Related Announcements
None

The total requests for new and amended subawards have steadily increased, per a recent update by Kim Carter, Executive Director, UK Office of Sponsored Projects Administration (OSPA). With over 400 requests in FY17, administering subawards has quickly become a major part of research administration operations both at the College of Medicine (COM) and University-wide levels. &nbs

By Vice President for Research Lisa Cassis Thursday
In May, it was my distinct pleasure to present Matthew Gentry with a University Research Professorship award. He was one of 16 faculty, selected by their own colleges, who have demonstrated excellence in scholarship and creative work that addresses scientific, social, cultural and economic challenges in our region and around the world.

Facilities and Administrative Cost (F&A) Rates
F&A Rate for Industry-funded Clinical Trials


New findings from University of Kentucky faculty published in Alzheimer’s Research and Therapy demonstrate the value of physician-pharmacist teams in providing elderly care.

NIH is working to improve the accuracy and reliability of its eRA Commons data. As part of this process, they are reviewing accounts with the intent to maintain only one account per investigator. eRA Commons allows investigators to have multiple roles within the same account, thus eliminating the need for duplicate accounts. Starting in late July, NIH will begin sending notifications to individuals who likely have more than one eRA Commons account. The contact will come in the form of an email which will provide instructions for selecting the preferred account.

A peek at Matthew Gentry's professional profile reveals a long list of honors and awards.
A National Science Foundation Faculty Career Development Award.A National Institutes of Health Pathway to Independence Award.A University of Kentucky Research Professorship Award.Three U.S. Patents.A five-year, $8.5 million National Institutes of Health grant to pursue a cure for Lafora disease.Wait. Lafora disease? From a lab that also studies biofuels?

NIH Fiscal Policy for Grant Awards - FY 2017
Notice Number: NOT-OD-17-086
Key Dates
Release Date: June 30, 2017

[from the NIH website]
Revision: Ruth L. Kirschstein National Research Service Awards (NRSA) Predoctoral Stipends, Training Related Expenses, Institutional Allowance, and Tuition/Fees Effective for Fiscal Year 2017
Notice Number: NOT-OD-17-084
Key Dates
Release Date: June 27, 2017